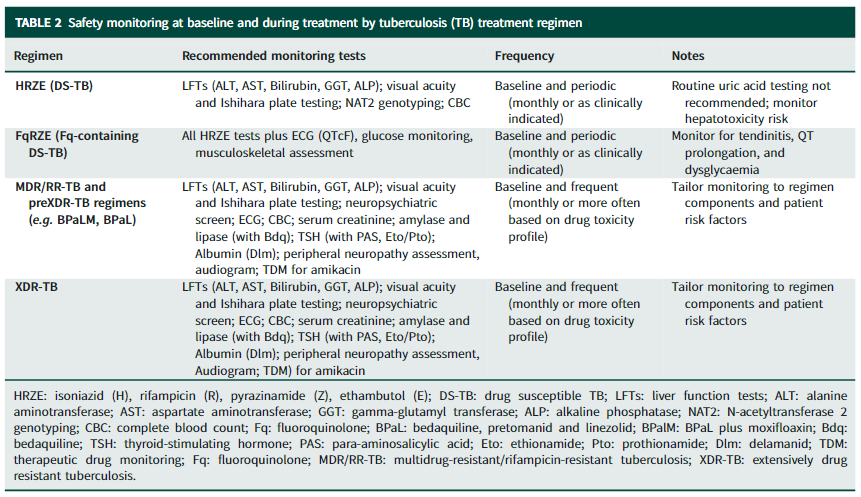
Abstract
Tuberculosis (TB) remains the leading cause of death from a single infectious agent, with 10.8 million cases reported in 2023. While treatment is generally effective, both drug-susceptible (DS-TB) and drug-resistant TB (DR-TB) regimens are associated with adverse events (AEs) that compromise adherence and outcomes. In this viewpoint, we highlight the most clinically relevant AEs: hepatotoxicity, cutaneous reactions and ocular toxicity in DS-TB; and linezolid-associated neuropathy, myelosuppression, QT prolongation and hepatotoxicity in DR-TB. We argue that structured safety monitoring and integration of active TB Drug Safety Monitoring and Management into routine care are critical to improving patient safety. Looking ahead, pharmacogenomics, therapeutic drug monitoring, predictive algorithms and digital health solutions offer opportunities to move from reactive to proactive AE management. By prioritising monitoring, innovation and patient-centred approaches, TB programmes can reduce the burden of AEs, improve adherence and achieve better outcomes.














